Materials and Methods
Staining protocols to quantify the number of viable pollen grains in a sample were adapted from a method published by V. Pinillos and J. Cuevas, which used fluorescein diacetate (FDA)(1). This method serves as an indicator of membrane integrity since only viable pollen grains fluoresce. Briefly, pollen was harvested from freshly cut anthers from the Datura stramonium flower. Stock FDA dissolved in DMSO was diluted to a concentration of 6 X 10-5 M in 0.5 M sucrose buffer. A clean pipette tip was used to transfer the pollen grains from the anther into 20 µL of FDA working solution and incubated for 5 minutes.
10 µL of sample was loaded onto the sample surfaces, and proper exposure was set to minimize saturation of the fluorescing pollen. Automated counting of pollen grains was performed according to the parameters in Table 1.
CellDrop EasyApps™ software is designed to count a variety of cell types with minimal customization. If necessary, the user can optimize count settings. Irregular Cell Mode may be selected to optimize counts for pollen species that are more elliptical or elongated.
Pollen Protocol Settings
Table 1: Count Settings for D. Stramonium pollen grains
| Count Application | AO |
| Chamber Height | 100 µm |
| Dilution Factor | 1 |
| Diameter (min) | 50 µm |
| Diameter (max) | 75 µm |
| Roundness | 1 |
| Green Fluorescence Threshold | 1 |
Results and Summary
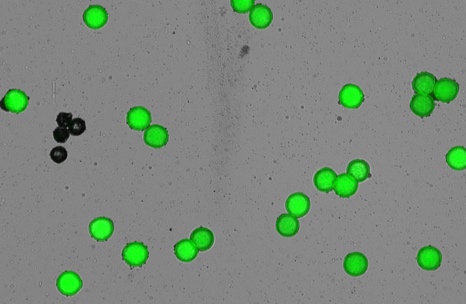
Viable pollen grains fluoresce green when FDA enters healthy pollen and is hydrolyzed by intracellular esterases (Figure 1). Non-viable pollen grains and debris do not fluoresce and are not counted. Results for total viable pollen counts are displayed as Live Cell Counts, and viable pollen concentrations are reported as Live Cells/mL.
The DeNovix CellDrop FL Automated Cell Counter, used in conjunction with FDA, increases efficiency and accuracy when determining viable pollen counts. Samples between 7 x 10^2 and 2.5 x 10^7 cells/mL and between 4 and 400 µm can be measured without dilution. The CellDrop EasyApps software uses powerful and flexible counting algorithms that allow users to customize count settings, enabling accurate counting and viability determination of various pollen types and sizes with rapid results.
1. V Pinillos & J Cuevas (2008) Standardization of fluorochromatic reaction test to assess pollen viability, Biotechnic & Histochemistry, 83:1, 15-21.
24-MAR-2025