Kit Contents
Three assay sizes are available. The volume of components in each kit are sufficient for 1000, 250 and 50 (evaluation size) assays respectively. Kit components are shown below. Safety data sheets are available at denovix.com/sds.
Table 1: Ultra High Sensitivity Assay Kit Contents| Component | 1000 | 250 | EVAL |
|---|---|---|---|
| DeNovix dsDNA Ultra High Sensitivity Dye (400x) | 0.5 mL | 125 µL | 25 µL |
| DeNovix dsDNA Ultra High Sensitivity Buffer | 200 mL | 50 mL | 10 mL |
| DeNovix dsDNA B Ultra High Sensitivity Enhancer (100x) | 2 x 1 mL | 0.5 mL | 100 µL |
| 300 pg/µL dsDNA Standard (calf thymus) | 2 mL | 1 mL | 0.5 mL |
| 0 pg/µL dsDNA Standard | 2 mL | 1 mL | 0.5 mL |
The dye is a potentially harmful chemical. Exercise universal laboratory safety precautions when handling the dye, and dispose of the dye as hazardous chemical waste according to your local regulations.
Instrument Compatibility
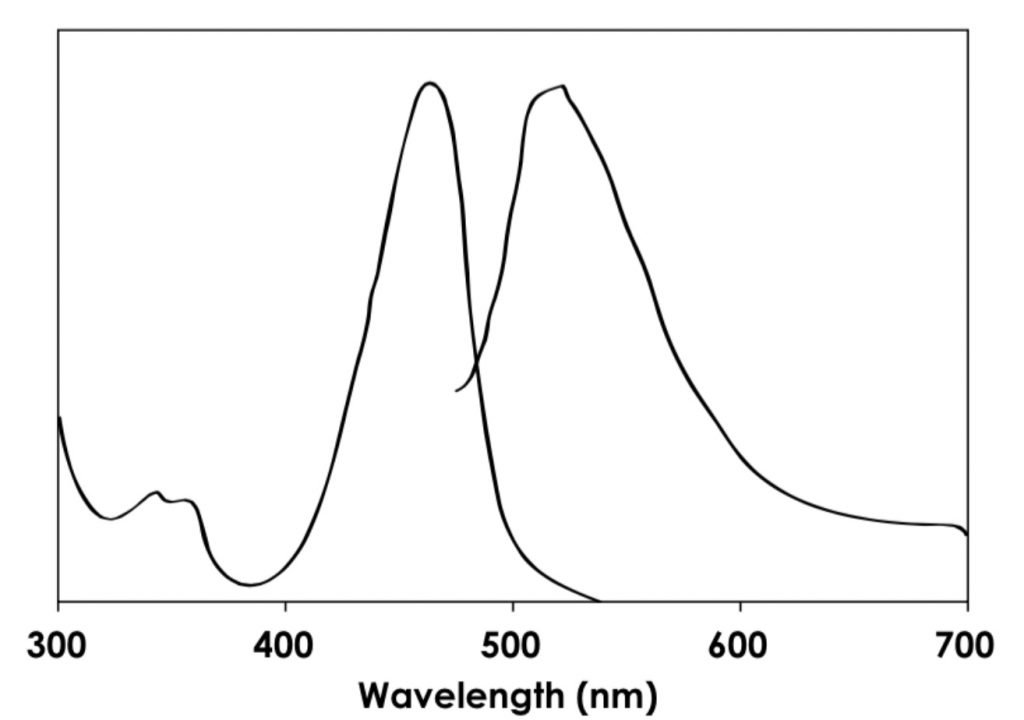
The DeNovix dsDNA High Sensitivity Quantitation Assay is designed for use with fluorometers or fluorescence plate readers equipped with excitation and emission filters for detecting green fluorescence. The unique spectral properties of the kit dye make it especially well-suited for use with instruments with blue LED excitation sources.
Specific instructions using the 2 Point Standard Assay with DeNovix DS-11 FX, FX module or the QFX fluorometer are included in Technical Note 146.
Assay Considerations
Calf thymus DNA is provided as the reference standard as it is double-stranded, highly polymerized and is approximately 58% AT (42% GC). At times it may be preferable to use a dsDNA standard similar to the unknown samples (e.g. similar in size, linear vs circular). For bacterial DNA, consider using a species-specific standard, as the GC content varies widely depending on the species.
Although many instruments, including the DeNovix DS-11 FX and QFX Fluorometers, offer the option to use previously saved values, it is recommended that a new standard curve be generated at the time of the assay for optimal results.
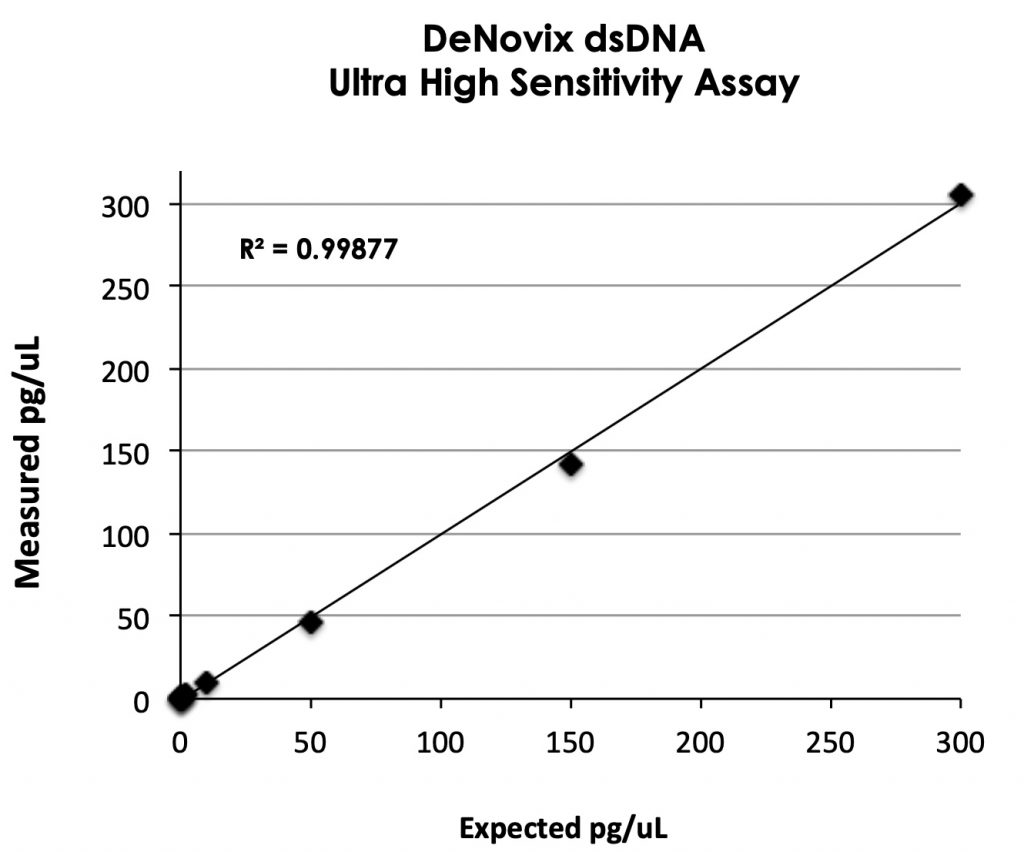
Assay Linearity and Detection Limits
Fluorescent quantification specifications are often expressed in a variety of conventions. The linear range of this assay can be expressed in the following equivalent specifications:
Table 2: Ultra High Sensitivity Linearity and Detection| Specification | Range |
|---|---|
| Absolute mass per assay tube | 5 pg to 3000 pg per 200 µL |
| Concentration in sample stock tube | 0.5 pg/µL to 300 pg/µL |
Note: These ranges are determined using the DeNovix FX Fluorometer. Your results may vary as a function of the sensitivity and accuracy of the instrument used.
Reagent Storage
The kit is stable for 12 months from ship date when stored as recommended.
Table 3: Ultra High Sensitivity Assay Reagent Storage| Component | Protect from Light | Temperature |
|---|---|---|
| DeNovix dsDNA Ultra High Sensitivity Dye (400x) | Yes | 4oC - Room Temperature |
| DeNovix dsDNA Ultra High Sensitivity Buffer | Optional | 4oC - Room Temperature |
| DeNovix dsDNA UltraHigh Sensitivity Enhancer (100x) | Optional | 4oC - Room Temperature |
| dsDNA Standards | Yes | 4oC |
Best Practices
It is important to pay careful attention to pipetting accuracy and overall sample handling techniques when quantitating picogram amounts of dsDNA.
Assay Protocol
- Allow all solutions to equilibrate to room temperature before use.
- Prepare 200 µL of working solution for each standard and sample to be tested by diluting the dye 1:400 in the assay buffer. Dilute the enhancer solution 1:100 into the dye/buffer mixture. Mix well before use.
- For each standard or unknown sample, add 200 µL of the working solution per tube or micro well.
- Add 10 µL of each standard or unknown DNA sample to the tube or micro well and mix well.
- Avoid introducing air bubbles when mixing samples and working reagent.
- Incubate standards and samples at room temperature for 5 minutes.
- Generate the standard curve and then measure the samples using the proper excitation source and emission filters.
Recommended Sample Volume
These recommendations ensure that sample concentrations are within the total mass detection limits of the assay.
| Initial Sample Concentration | Recommended Sample Volume |
|---|---|
| 0.5 – 300 pg/µL | 10 µL |
Standard Dilutions
Standard Dilutions Preparing diluted standards is not required when using the 2 point assay option supplied. For the DeNovix User Defined Standards option or for use on microplate readers, prepare DNA standards by serial dilution of the 300 pg/µL standard in the 1X assay buffer as shown in the table below.
| Standard | DNA | TE |
|---|---|---|
| 300 pg/µL | 100 µL of 300 pg/µL stock tube | None |
| 150 pg/µL | 100 µL of 300 pg/µL standard | 100 µL |
| 50 pg/µL | 75 µL of 150 pg/µL standard | 150 µL |
| 10 pg/µL | 40 µL of 50 pg/µL standard | 160 µL |
| 2 pg/µL | 40 µL of10 pg/µL standard | 160 µL |
| 1 pg/µL | 100 µL of 2 pg/µL standard | 100 µL |
| 0.5 pg/µL | 100 µL of1 pg/µL standard | 100 µL |
| 0 pg/µL | 100 µL of 0 ng/µL stock tube | None |
Data Analysis
Sample concentrations are automatically calculated when using a DeNovix DS-11 FX or QFX Fluorometer.
For all other instruments, follow the instructions below:
- Generate a standard curve to determine the unknown DNA concentration.
- Average replicate values for each sample and subtract the average zero DNA value from each data point.
- Plot the fluorescence RFU values for the DNA standards on the y-axis and pg/well DNA on the x-axis, and fit a trend line (Figure 2) through these points to generate a standard curve with a y-intercept = 0.
- Use the equation for the trend line to calculate the amount of unknown DNA in each well (y = fluorescence and x = pg DNA per well or tube).
Troubleshooting
Appendix: Solvent Compatibility
Table 5: Ultra High Sensitivity Assay Appendix| Compound | Final concentration in assay (200 uL) | Signal Decrease (%) |
|---|---|---|
| Sodium Chloride | 25 mM | 7% |
| Sodium Acetate | 30 mM | 11% |
| Magnesium Chloride | 5 mM | 30% |
| SDS | 0.01% | 87% |
| SDS | 0.001% | 13% |
| Ethanol | 1% | 8% |
| Phenol | 0.10% | 10% |
| Triton X-100 | 0.01% | 18% |
| Triton X-100 | 0.001% | 8% |
| Tween-20 | 0.005% | 8% |
| CTAB* | 0.0005% | 100% |
| dNTPs | 100 uM | 1% |
| BSA** | 0.04 mg/mL | 19% |
* CTAB generally inhibits fluorescence, but at 5 pg the background is increased.
** BSA is not compatible with quantitation below 500 pg due to increased background at low DNA concentrations.
DeNovix Assays
If the Ultra High Sensitivity Assay does not cover the concentration range of your samples, consider using an alternate DeNovix dsDNA Assay Kit.
For comparison, the standard detection ranges of the three assays are as follows:
Assay Detection Ranges| DeNovix dsDNA Assay | Range |
|---|---|
| Broad Range | 0.1 – 2000 ng/µL (extended range to 4000 ng/uL) |
| High Sensitivity | 10 pg/µL – 250 ng/µL (extended range down to 5 pg/uL) |
| Ultra High Sensitivity | 0.5 – 300 pg/µL |
Summary
Contact DeNovix Customer Support if further help is required. Outside of the US, please contact your local distributor for assistance.
For instructions specific on performing a 2 Point Standard Curve Assay on a DeNovix Fluorometer, refer to Technical Note 146 – Ultra High Sensitivity Assay Standard Protocol.
10-JAN-2025