How Accurate is the DeNovix Squid Pipette?
The DeNovix Squid Pipette demonstrates accuracy and precision across its entire 1 – 1000 µL volume range. Squid meets or exceeds ISO 8655 requirements, as proven by the data in this technical note, and therefore offers a reliable alternative to conventional pipette sets.
Abstract
The Squid Pipette is a novel electronic, single-channel, air-displacement pipette with a continuously adjustable volume range of 1 to 1000 µL. This technical note presents performance validation data demonstrating compliance with ISO 8655-2:2022 across the complete volume range. Testing was conducted in accordance with the gravimetric reference procedure specified in ISO 8655:2022 under controlled environmental conditions.
Results demonstrate that the Squid Pipette meets or exceeds all maximum permissible error limits (MPE) for both systematic error (trueness) and random error (precision) at all test volumes. These data support the use of the Squid as a direct replacement for a conventional pipette set (typically P10, P20, P200, and P1000), reducing equipment cost, bench space, and calibration overhead without compromising accuracy.
1. Introduction
1.1 Background
Pipetting volumes between 1 and 1000 µL form the majority of liquid handling steps in the modern life science laboratory. Traditionally, between three and five separate pipettes are needed to accurately span this working range. Each instrument must be prominently stored on the lab bench as well as independently calibrated, maintained, and verified. This creates a significant operational burden in both cost and time.
The Squid Pipette has been engineered to cover this entire range within a single instrument. Built on the renowned expertise of DeNovix in microvolume motor control and a patented dual tip system, the device maintains accuracy and precision across its full 1 – 1000 µL range and at both extremes.
1.2 Regulatory Context: ISO 8655:2022
ISO 8655 is the internationally recognized standard governing the calibration and performance testing of piston-operated volumetric apparatus (POVA), including all single- and multi-channel pipettes. The 2022 revision introduced several material changes relevant to wide-range instruments:
- Error limits are now defined at three nominal volume fractions: 10%, 50%, and 100% of the nominal volume — making compliance at extreme ends of the range a formal requirement.
- The pipette and its tip(s) are treated as a single integrated system; calibration is valid only for the specific tip type used during testing.
- A minimum of 10 measurements per test volume is required, across at least 3 volumes, with at least one tip change during testing.
- Balance requirements have been tightened: a 6-place balance is required for volumes ≤20 µL; a 5-place balance for volumes between 20 µL and 199 µL.
Compliance with ISO 8655:2022 therefore constitutes a stringent, internationally benchmarked demonstration of performance, providing customers with an objective and standardized basis for evaluation.
1.3 Purpose of This Technical Note
This document presents:
- The test protocol used for validation, including equipment specifications and environmental conditions.
- Raw and processed performance data at multiple volumes across the full 1 – 1000 µL range.
- Direct comparison of Squid performance against ISO 8655:2022 maximum permissible errors (MPEs).
2. Materials and Methods
2.1 Test Instruments
Table 1. Device Parameters
| Parameter | Specification |
|---|---|
| Instrument | Squid Pipette |
| Volume Range | 1 - 1000 µL (Continuously Adjustable) |
| Displacement Type | Air-Displacement (Type A, per ISO 8655-2) |
| Channel Configuration | Single-Channel |
| Tip Type Used | DeNovix 200 µL Sterile Tips DeNovix 1000 µL Sterile Tips |
| Device Serial Numbers Tested | A99983, A99998, A99990 |
2.2 Test Volumes
The current ISO 8655 standard is understandably designed to test traditional, narrow range pipettes. The standard recommends systematic and random error values specific to each pipette range at 100%, 50% and 10% of the nominal maximum dispense volume. These parameters are unsuitable for the Squid Pipette as it would only require testing at 1000, 500 and 100 µL.
To comply with the spirit of the regulations, ensure data quality, and verify the claim that Squid replaces a full range of pipettes without compromising quality, the testing parameters were expanded in this study to include all of the testing volumes and ISO Maximum Permitted Errors (MPE) for four conventional range pipettes (P10 to P1000).
Test volumes were selected to satisfy the ISO 8655:2022 requirement of measurements at 100%, 50% and 10% of nominal volume. Squid utilizes a patented dual tip mechanism compatible with both 200 and 1000 µL tips. Testing was performed using Small Tip Mode in the range of 1 – 200 µL and Large Tip Mode in the range of 100 to 1000 µL.
The following volumes were tested:
Table 2.Target Volumes Tested Using Squid
| Test Volume (µL) | % of 1000 µL Nominal | Analogous Conventional Pipette |
|---|---|---|
| 1 | 0.1% | P10 10% |
| 2 | 0.2% | P20 10% |
| 5 | 0.5% | P10 50% |
| 10 | 1% | P10 100% P20 50% |
| 20 | 2% | P20 100% P200 10% |
| 100* | 10% | P200 50% P1000 10% |
| 200 | 20% | P200 100% |
| 500 | 50% | P1000 50% |
| 1000 | 100% | P1000 100% |
*Performed using both 200 µL and 1000 µL pipette tips
Table 3. Maximum Permitted Error Values for Standard Pipettes Defined by ISO 8655
| Pipette Nominal Range (µL) | Nominal Volume (µL) | ISO MPE Systematic Error - % CV | ISO MPE Random Error- % Error |
|---|---|---|---|
| 1 - 10 | 1 | 8.0 | 12.0 |
| 5 | 1.6 | 2.4 | |
| 10 | 0.8 | 1.2 | |
| 2 - 20 | 2 | 5.0 | 10.0 |
| 10 | 1.0 | 2.0 | |
| 20 | 0.5 | 1.0 | |
| 20 - 200 | 20 | 3.0 | 8.0 |
| 100 | 0.6 | 1.6 | |
| 200 | 0.3 | 0.8 | |
| 100 - 1000 | 100 | 3.0 | 8.0 |
| 500 | 0.6 | 1.6 | |
| 1000 | 0.3 | 0.8 |
2.3 Gravimetric Test Procedure (ISO 8655-6:2022)
All measurements were performed using the gravimetric reference procedure defined in ISO 8655-6:2022. The procedure is summarized below.
Table 4. Equipment
| Equipment | Specification | Purpose |
|---|---|---|
| Analytical Balance Equipped with Environmental Sensor | 6-Place (d = 0.001 mg) for Volumes ≤20 µL | Mass Measurement |
Table 5. Environmental Conditions of Study
| Parameter | ISO 8655 Requirement | Actual Conditions (Mean ± SD) |
|---|---|---|
| Laboratory Temperature | 15–30 °C (Stable within ±0.5 °C) | 22.6 °C |
| Water Temperature | 15–30 °C (Reported to 0.1 °C) | 22.1 °C |
| Relative Humidity | 45–75% RH | 52.8% |
| Atmospheric Pressure | Recorded for Z-Factor Correction | 100.3 kPa |
2.3.1 Pre-Test Preparation
- Pipette and tips equilibrated to laboratory temperature for ≥2 hours prior to testing.
- Tips pre-wetted with 3 aspirate-dispense cycles at the test volume before data collection.
- Tips changed at least once per volume tested (ISO 8655-6:2022 requirement).
- Balance zeroed and verified with reference weights before each volume series.
2.3.2 Measurement Protocol
- 10 replicate measurements per test volume (ISO 8655-6:2022 minimum).
- Liquid dispensed into a covered vessel on the balance; evaporation monitored throughout.
- Mass values recorded to full balance resolution.
- Three independent, randomly selected Squid units tested to assess unit-to-unit manufacturing variation.
3. Results
Volume, systematic error and random error data are presented for the three Squid devices under study and compared to the Maximum Permitted Errors defined by ISO 8655. Data are presented for a representative device from the three under study in addition to a comparison of all three devices to demonstrate manufacturing consistency.
3.1 Performance Summary Table
The performance data across all test volumes is presented in table 6. Volume values are the mean of ten replicates. ISO pass / fail criteria are based on appropriate Mean Permitted Errors for the most appropriate standard pipette for delivering the specified volume. Values are reported for a representative single unit (S/N A99990); multi-unit data are presented in Section 3.3.
Table 6. Performance Validation Results for the Squid Pipette Across its Full Operating Range (1 – 1000 µL)
Data represents mean delivered volumes, systematic error (accuracy), and random error (precision) as compared against ISO 8655:2022 permissible limits.
| Nominal Volume (µL) | Tip Size | Mean Delivered (µL) | Systematic Error (%) | Random Error (CV%) | ISO Standard Pass/Fail |
|---|---|---|---|---|---|
| 1 | 200 µL | 0.962 | 3.834 | 7.133 | ✅ |
| 2 | 200 µL | 1.945 | 2.750 | 3.416 | ✅ |
| 5 | 200 µL | 5.063 | 1.259 | 1.450 | ✅ |
| 10 | 200 µL | 10.025 | 0.258 | 0.633 | ✅ |
| 20 | 200 µL | 19.999 | 0.128 | 0.411 | ✅ |
| 100 | 200 µL | 99.947 | 0.053 | 0.222 | ✅ |
| 200 | 200 µL | 200.516 | 0.258 | 0.172 | ✅ |
| 100 | 1000 µL | 100.286 | 0.286 | 0.261 | ✅ |
| 500 | 1000 µL | 498.617 | 0.277 | 0.181 | ✅ |
| 1000 | 1000 µL | 998.487 | 0.151 | 0.153 | ✅ |
3.2 Precision (Random Error / CV) and Accuracy (Systematic Error / % Error) Across the Full Volume Range
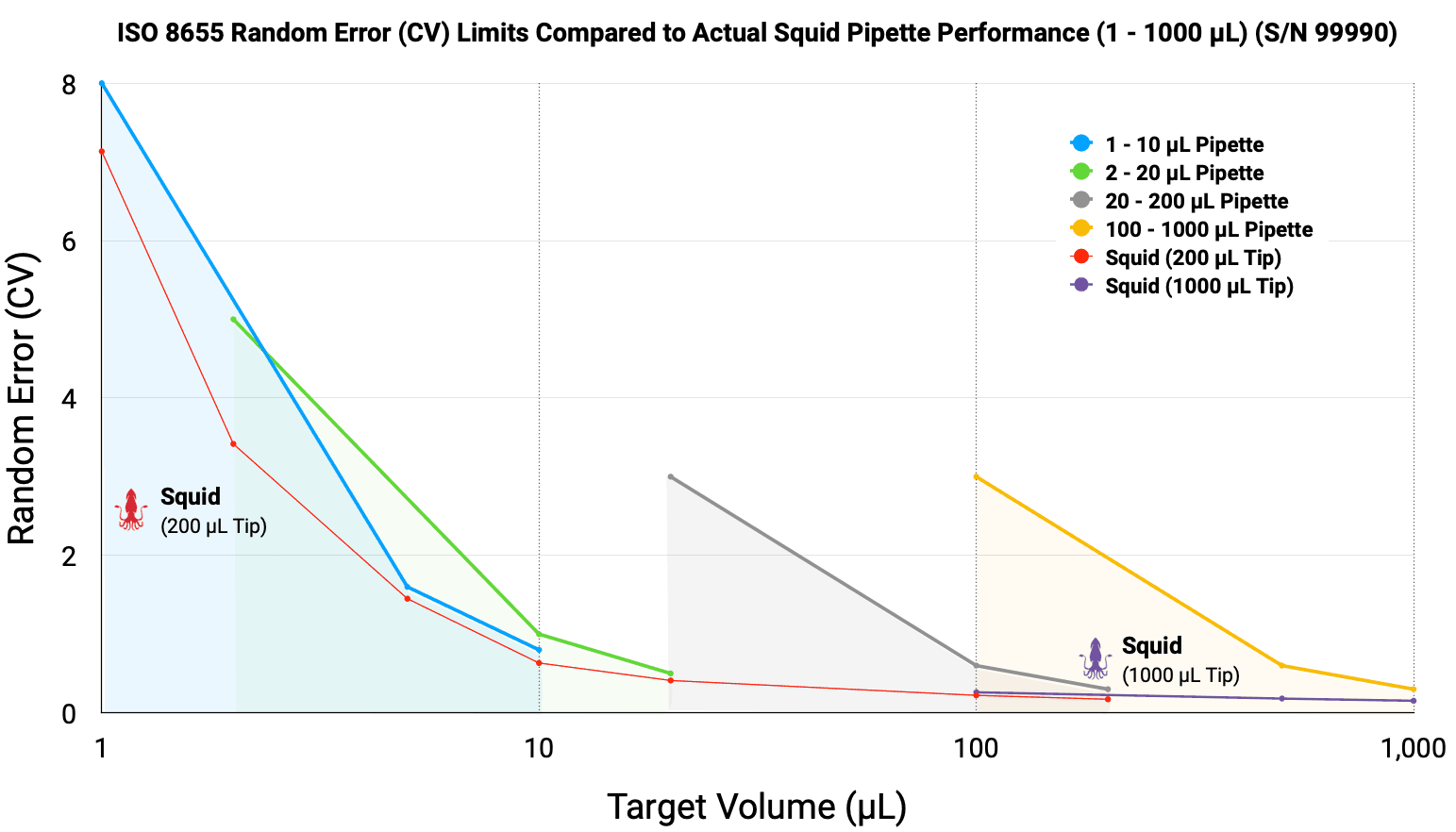
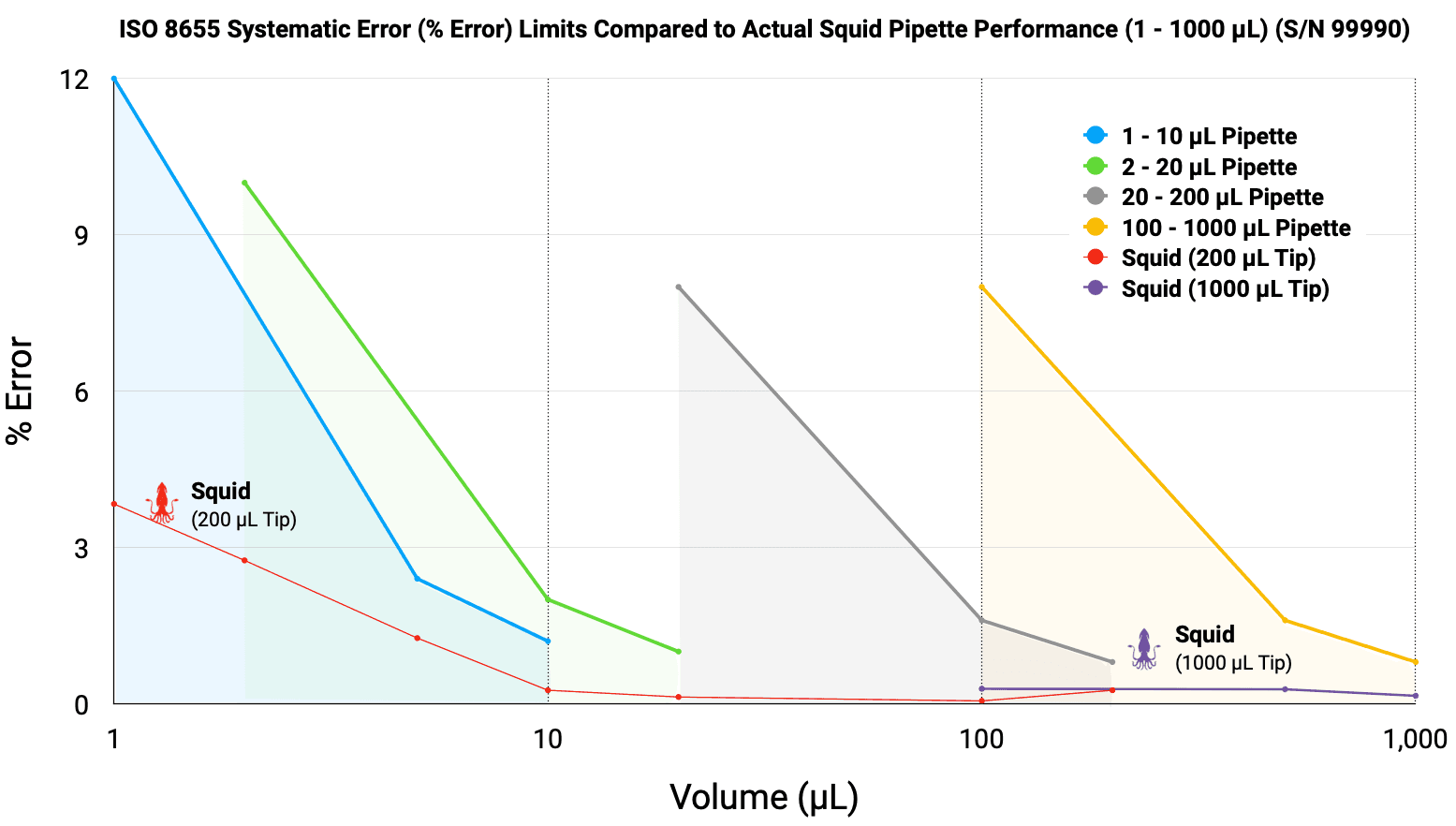
The operational performance of the Squid Pipette is demonstrated by evaluating its performance against the maximum permissible errors defined by ISO 8655:2022. Traditional air-displacement pipetting typically requires a suite of four distinct instruments (1 – 10 µL, 2 – 20 µL, 20 – 200 µL, 100 – 1000 µL) to cover the volume. Squid was evaluated against the recommended Maximum Permitted Error for each of these. As illustrated in the figures below, the Squid exceeds the ISO requirements for precision and accuracy across the full volume range tested.
Figure 1. Random Error (CV) comparison between the Squid Pipette (S/N 99990) and ISO 8655 permissible limits. The Squid Pipette demonstrates superior precision across the entire 1 – 1000 µL range, consistently performing well below the maximum allowable limits for standard air-displacement pipettes.
Figure 2. Systematic Error (% Error) of the Squid Pipette (S/N 99990) compared to ISO 8655:2022 permissible limits. The Squid performance (red and purple lines) remains consistently below the maximum allowable limits across all volumes, demonstrating superior accuracy. The overlapping data points at 100 µL and 200 µL illustrate the seamless transition and maintained performance between the 200 µL and 1000 µL tip types.
3.3 Unit-to-Unit Reproducibility (Manufacturing Consistency)
To assess the consistency of performance across different physical units, the between-unit reproducibility was calculated. This analysis evaluates the variation between the mean volumes of three separate Squid Pipette units across the full volume range.
The results demonstrate extremely high manufacturing and calibration consistency. For all volumes 5 µL and above, the between-unit CV remains below 1%, indicating excellent uniformity across the product line. At the lowest volumes (1 µL and 2 µL), while the relative Coefficient of Variation (CV%) appears higher at 4.39% and 3.87% respectively, it is important to note that this represents an extremely small absolute deviation—less than 0.08 µL. Furthermore, the Grand Mean for the 1 µL setting was 0.987 µL, demonstrating that the actual volume delivered remains remarkably close to the nominal target.
Table 7. Between-Unit Reproducibility Analysis (Grand Mean, SD, and CV%) for Squid Pipette Units Across Nine Nominal Volume Setpoints
| Volume (µL) | Average Grand Mean (μL) | Average Between-Unit SD | Average Between-Unit CV (%) |
|---|---|---|---|
| 1 | 0.987 | 0.0433 | 4.39% |
| 2 | 2.0327 | 0.0786 | 3.87% |
| 5 | 5.066 | 0.0436 | 0.86% |
| 10 | 9.9773 | 0.0667 | 0.67% |
| 20 | 19.9532 | 0.0648 | 0.32% |
| 100 | 100.0447 | 0.5429 | 0.54% |
| 200 | 201.003 | 0.5352 | 0.27% |
| 500 | 500.065 | 1.5447 | 0.31% |
| 1000 | 1001.8703 | 2.9683 | 0.30% |
Note: Grand Mean represents the average of the mean volumes delivered by three separate units (A99983, A99998, and A99990).
4. Conclusion
The validation data across Units A99983, A99998, and A99990 confirms that the Squid Pipette performs reliably within the rigorous framework of ISO 8655:2022 standards. By maintaining a high degree of linear accuracy and precision across a dynamic range of 1 µL to 1000 µL, the system effectively consolidates the capabilities of multiple traditional air-displacement pipettes into a single, versatile platform.
A defining characteristic of the Squid device is its performance stability. While ISO regulations accommodate an increasing error for regular pipettes as they approach their lower volumetric limits, the Squid Pipette maintains low error at every ISO setpoint. The higher relative CV% observed at the 1 µL and 2 µL levels is a mathematical consequence of the micro-scale volumes; in absolute terms, the volumetric deviation remains negligible (less than 0.08 µL, and the delivered means remain remarkably congruent with nominal targets.
Furthermore, the between-unit reproducibility study highlights the consistency of the Squid manufacturing process. With an average between-unit CV of approximately 0.29 µL at higher volumes and consistent micro-volume delivery across all tested units, researchers can move between different instruments with total confidence in data interchangeability. This minimizes the “instrument variable” in complex workflows, ensuring that results are reproducible regardless of the specific unit utilized.
In conclusion, the Squid Pipette represents a robust, high-precision solution that exceeds international standards. Its ability to provide consistent, accurate, and precise liquid handling across three orders of magnitude validates it as a primary tool for demanding laboratory environments, including GLP-compliant research and clinical diagnostics.
Ready to Join the #SquidSquad?
Your Squid Pipette is waiting for you. Buy now or request a quote to start simplifying your workflow.
16-APR-2026